Studies of atomic-thickness thin films (2D) have been at the forefront of research in recent decades. A large body of information has been gathered primarily on metals and thin oxide layers, which resulted in a number of technologies in corrosion protection, sensors, and catalysis.
Strictly speaking, the beginning of the 21st century is the birth of a discipline, based on the experience gained in the above-mentioned thin films, that aims to study surface structures that include conductors, semiconductors, and insulators.
Our model studies are performed in a research group unique in Hungary, where in addition to determining the atomic-scale structure and morphology of catalytically active surfaces (STM, SEM, TEM, XRD), we can also perform their chemical and elemental analysis (XPS, AES, LEIS). This is complemented by the vibrational spectroscopic characterization of surface adsorbates and supports (FTIRS, DRIFTS, HREELS, Raman) as well as the analysis of gas-phase reaction products of thermal catalysis (MS, TDS, GC). The experimental results are supplemented with theoretical calculations.
The most important scientific results of the research group

In addition to the study of BN layers, successful research was conducted on the growth mechanism, oxidation state and structural changes of thin oxide films (TiO2, CeO2, MoO2) formed on different substrates (Rh, TiO2, Cu). The characterized surfaces were modified with different metals (Mo, Au, Co), and the structural changes due to temperature were monitored by surface science techniques. The interactions between the adsorption sites on the surface and the CO and CO2 molecules were examined through spectroscopic techniques. The results obtained here contribute to a deeper understanding of the catalytic transformation of these molecules.
In addition to the 2D layers, we studied the structure of rhodium-gold nanoparticles (0D) formed on oxides and their activity in the photocatalytic conversion of CO2 and CH4.
Results of the last 1 year
The primary focus of the research group is the preparation and investigation of low dimensional (0D, 1D and 2D) materials, showing promising physical and chemical properties unprecedented in the bulk phase. The efforts of research group are devoted to the investigation of the elementary steps of surface reactions and processes, which have a significant role in practically applicable nanotechnological processes.
Farkas AP, Szitás Á, Jurdi D, Palotás K, Kiss J, Kónya Z. Applied Catalysis A: General. 2020 Feb 25;592:117440. https://doi.org/10.1016/j.apcata.2020.117440
Szitás Á, Gubó R, Pásztor T, Farkas AP, Ajtai T, Óvári L, Palotás K, Berkó A, Kónya Z. The Journal of Physical Chemistry C. 2020 Jun 3;124(26):14182-94. https://doi.org/10.1021/acs.jpcc.0c01725
The 5 most important publications of the research group
-
Farkas, AP; Szitás, Á ; Jurdi, D ; Palotás, K. ; Kiss, J ; Kónya, Z
Selective transformation of ethanol to acetaldehyde catalyzed by Au/h-BN interface prepared on Rh(111) surface
APPLIED CATALYSIS A-GENERAL 592 Paper: 117440 , 9 p. (2020) https://doi.org/10.1016/j.apcata.2020.117440 -
Szitas, A ; Gubo, R ; Pasztor, T ; Farkas, AP ; Ajtai, T ; Ovari, L ; Palotas, K ; Berko, A ; Konya, Z
Adsorption of Azobenzene on Hexagonal Boron Nitride Nanomesh Supported by Rh(111)
JOURNAL OF PHYSICAL CHEMISTRY C 124 : 26 pp. 14182-14194. , 13 p. (2020) https://doi.org/10.1021/acs.jpcc.0c01725 -
Varga, T ; Ballai, G ; Vásárhelyi, L ; Haspel, H ; Kukovecz, Á ; Kónya, Z
Co4N/nitrogen-doped graphene: a non-noble metal oxygen reduction electrocatalyst for alkaline fuel cells
APPLIED CATALYSIS B-ENVIRONMENTAL 237 pp. 826-834. (2018) https://doi.org/10.1016/j.apcatb.2018.06.054 -
Gubo, R ; Vari, G ; Kiss, J ; Farkas, AP ; Palotas, K ; Ovari, L ; Berko, A ; Konya, Z
Tailoring the hexagonal boron nitride nanomesh on Rh(111) with gold
PHYSICAL CHEMISTRY CHEMICAL PHYSICS 20 : 22 pp. 15473-15485. , 13 p. (2018) https://doi.org/10.1039/C8CP00790J -
Palotás, K ; Óvári, L ; Vári, G ; Gubó, R ; Farkas, AP ; Kiss, J ; Berkó, A ; Kónya, Z
Au–Rh Surface Structures on Rh(111): DFT Insights into the Formation of an Ordered Surface Alloy
JOURNAL OF PHYSICAL CHEMISTRY C 122 : 39 pp. 22435-22447. , 13 p. (2018) https://doi.org/10.1021/acs.jpcc.8b05744
Head of the research group: Prof. Zoltán Kónya

Prof. Zoltán Kónya co-authored over 400 peer-reviewed papers, 15 book chapters and holds 13 patents; he has over 8500 independent citations and his H-index is 52.
The most important tools
Available characterization techniques: ultra high vacuum equipments/techniques (Auger electron spectroscopy (AES), Temperature Programmed Reaction/Desorption (TPR/TPD), Ultraviolet- and X-ray photoelectron spectroscopy (UPS-XPS), Low-energy ion scattering spectroscopy (LEIS), High resolution electron energy loss spectroscopy (HREELS), scanning tunneling microscope (STM)), chromatography (GC-MS, HPLC-MS), electron microscopy (HRTEM, FESEM), atomic force microscopy (AFM), Raman- and FTIR microscopy, etc.
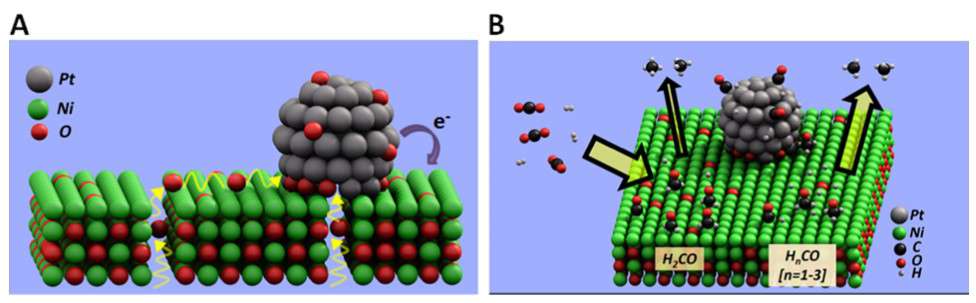
The most important figures connected to the research fields

Industrial partners
- Auro-science Kft. https://auroscience.hu/
- OKFT Kft. https://www.okft.hu/
- MOL https://mol.hu/hu/
- Hungarotex Zrt. http://hungarotex.eu/sajat_projektek.html
- Ganz Energetikai és Technológiai Kft. http://www.ganztech.eu/
Contact the research group leader on the following e-mail address:
konya@chem.u-szeged.hu
